Subject: TRI
Comprehensive Review Highlights Potential Benefits of Leukine® in Management of Gastrointestinal (GI) Adverse Events Associated with Immune Checkpoint Inhibitors (ICIs) in the Treatment of Cancer
NCI sponsored, ECOG-ACRIN Phase 3 Study of Leukine in Combination with Ipilimumab and Nivolumab in Front Line Treatment of Melanoma Heads to Last Stages of Enrollment
LEXINGTON, Mass., April 18, 2024 /PRNewswire/ -- Partner Therapeutics, Inc. (PTx) announced publication of a comprehensive review by Dougan et al.1 in Cancers summarizing the mechanistic rationale and pre-clinical and clinical data regarding the use of Leukine (sargramostim; glycosylated, yeast-derived recombinant human granulocyte-macrophage colony-stimulating factor [rhu GM-CSF]) to manage gastrointestinal (GI) immune-related adverse events (irAEs) for patients taking immune checkpoint inhibitors (ICIs) for the treatment of cancer. Leukine is not FDA approved for treatment of irAEs or melanoma.
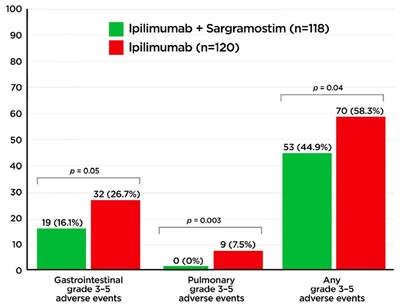
The authors describe the mechanisms underlying GI irAEs and their similarities to inflammatory bowel disease and review the available clinical data supporting a potential role for Leukine in both settings. In the prospective, randomized study E1608, Hodi and colleagues2 observed that advanced melanoma patients who received Leukine plus ipilimumab had a reduction in GI irAEs (p=0.05) (see figure below) as well as a significant improvement in overall survival (p=0.01) compared to those who received ipilimumab alone.
"Immune checkpoint inhibitors have meaningfully improved prognoses for cancer patients in a number of cancers," said Michael Dougan MD, PhD, Associate Professor of Medicine at Massachusetts General Hospital. "Unfortunately, these drugs also can cause the immune system to attack healthy cells creating unwanted GI side effects including some that lead to discontinuation of treatment and more severe outcomes. The potential Leukine has shown in mitigating GI irAEs and improving patient outcomes, including survival, are encouraging."
Ongoing Phase 3 Study of Leukine with Ipilimumab and Nivolumab in Front Line Metastatic Melanoma
Leukine continues to be studied in combination with ICIs to assess differences in the rate of serious adverse events as well as its impact on overall survival.
EA6141 (NCT02339571) is a randomized, controlled phase 2/3 study of Leukine in combination with ipilimumab and nivolumab for the front line treatment of patients with unresectable stage III or stage IV melanoma that is sponsored by the National Cancer Institute (NCI) and being conducted by ECOG-ACRIN Cancer Research Group.3 It is led by Study Chair F. Stephen Hodi Jr., MD, Director of the Center for Immuno-Oncology at Dana-Farber Cancer Institute, and Study Co-Chair Ahmad A. Tarhini, MD, PhD, Professor of Oncologic Sciences and Director of Cutaneous and Clinical Translational Research at H. Lee Moffitt Cancer Center and Research Institute. Both are members of the ECOG-ACRIN Melanoma Committee.
"The phase 2 portion of study EA6141 was completed previously, and after a successful planned interim analysis, advanced to the phase 3 portion, which is now in the last stages of enrollment," said Dr. Hodi in a recent statement. "The primary objective is to compare the overall survival rates of patients who receive nivolumab/ipilimumab/rhu GM-CSF versus those who receive only nivolumab/ipilimumab."
REFERENCES
- Dougan M, Nguyen LH, Buchbinder EI, Lazarus HM. Sargramostim for Prophylactic Management of Gastrointestinal Immune-Related Adverse Events of Immune Checkpoint Inhibitor Therapy for Cancer. Cancers. 2024; 16(3):501. doi.org/10.3390/cancers16030501
- Hodi FS, Lee S, McDermott DF, et al. Ipilimumab Plus Sargramostim vs Ipilimumab Alone for Treatment of Metastatic Melanoma: A Randomized Clinical Trial. JAMA. 2014; 312(17):1744-1753. doi:10.1001/jama.2014.13943
- National Cancer Institute. A Phase II/III Trial of Nivolumab, ipilimumab, and GM-CSF in patients with advanced melanoma. Available at: https://clinicaltrials.gov/ct2/show/NCT02339571. NLM identifier: NCT02339571. Accessed April 16, 2024.
ABOUT MELANOMA
Melanoma is the most aggressive form of skin cancer and rates of melanoma have been rising for the past 30 years. The American Cancer Society estimates 96,480 new melanoma cases will be diagnosed in the US and 7,230 people will die from the disease in 2019. The FDA grants orphan drug designation to promote the development of promising treatments for conditions that affect 200,000 or fewer U.S. patients annually.
ABOUT LEUKINE
LEUKINE (sargramostim) is a glycosylated recombinant human granulocyte-macrophage colony-stimulating factor (rhu GM-CSF) produced by recombinant DNA technology in yeast. The product is commercially available in the United States and accessible through a named patient program operated by Tanner Pharma Group outside of the United States.
LEUKINE is indicated:
- To shorten time to neutrophil recovery and to reduce the incidence of severe and life-threatening infections and infections resulting in death following induction chemotherapy in adult patients 55 years and older with acute myeloid leukemia (AML).
- For the mobilization of hematopoietic progenitor cells into peripheral blood for collection by leukapheresis and autologous transplantation in adult patients.
- For the acceleration of myeloid reconstitution following autologous bone marrow or peripheral blood progenitor cell transplantation in adult and pediatric patients 2 years of age and older.
- For the acceleration of myeloid reconstitution following allogeneic bone marrow transplantation in adult and pediatric patients 2 years of age and older.
- For treatment of delayed neutrophil recovery or graft failure after autologous or allogeneic bone marrow transplantation in adult and pediatric patients 2 years of age and older.
- To increase survival in adult and pediatric patients from birth to 17 years of age acutely exposed to myelosuppressive doses of radiation (Hematopoietic Syndrome of Acute Radiation Syndrome [H-ARS]).
Important Safety Information for Leukine (sargramostim)
Contraindications
- Do not administer LEUKINE to patients with a history of serious allergic reaction, including anaphylaxis, to human granulocyte-macrophage colony-stimulating factor, sargramostim, yeast-derived products, or any other component of LEUKINE.
Warnings and Precautions
- Serious hypersensitivity reactions, including anaphylactic reactions, have been reported with LEUKINE. If a serious allergic or anaphylactic reaction occurs, immediately discontinue LEUKINE therapy, and institute medical management. Discontinue LEUKINE permanently for patients with serious allergic reactions.
- LEUKINE can cause infusion-related reactions that may be characterized by respiratory distress, hypoxia, flushing, hypotension, syncope, and/or tachycardia. Observe closely during infusion, particularly in patients with preexisting lung disease; dose adjustment or discontinuation may be needed.
- LEUKINE should not be administered simultaneously with or within 24 hours preceding cytotoxic chemotherapy or radiotherapy or within 24 hours following chemotherapy.
- Edema, capillary leak syndrome, and pleural or pericardial effusions have been reported in patients after LEUKINE administration. LEUKINE should be used with caution in patients with preexisting fluid retention, pulmonary infiltrates, or congestive heart failure. Such patients should be monitored.
- Supraventricular arrhythmia has been reported in uncontrolled studies during LEUKINE administration, particularly in patients with a history of cardiac arrhythmia. Use LEUKINE with caution in patients with preexisting cardiac disease.
- If absolute neutrophil count (ANC) > 20,000 cells/mm3 or if white blood cell (WBC) counts > 50,000/mm3, LEUKINE administration should be interrupted, or the dose reduced by half. Monitor complete blood counts (CBC) with differential twice per week.
- Discontinue LEUKINE therapy if tumor progression, particularly in myeloid malignancies, is detected during LEUKINE treatment.
- Treatment with LEUKINE may induce neutralizing anti-drug antibodies. Use LEUKINE for the shortest duration needed.
- Avoid administration of solutions containing benzyl alcohol (including LEUKINE for injection reconstituted with Bacteriostatic Water for Injection, USP [0.9 % benzyl alcohol]) to neonates and low birth weight infants.
Drug Interactions
- Avoid the concomitant use of LEUKINE and products that induce myeloproliferation. Monitor for clinical and laboratory signs of excess myeloproliferative effects.
Adverse Reactions
Adverse events occurring in >10% of patients receiving LEUKINE in controlled clinical trials and reported at a higher frequency than in placebo patients are:
- In recipients of autologous bone marrow transplantation (BMT)?asthenia, malaise, diarrhea, rash, peripheral edema, urinary tract disorder
- In recipients of allogeneic BMT?abdominal pain, chills, chest pain, diarrhea, nausea, vomiting, hematemesis, dysphagia, GI hemorrhage, pruritus, bone pain, arthralgia, eye hemorrhage, hypertension, tachycardia, bilirubinemia, hyperglycemia, increased creatinine, hypomagnesemia, edema, pharyngitis, epistaxis, dyspnea, insomnia, anxiety, high glucose, low albumin
- In patients with AML?fever, weight loss, nausea, vomiting, anorexia, skin reactions, metabolic laboratory abnormalities, edema
ABOUT PARTNER THERAPEUTICS
Partner Therapeutics, Inc. (PTx), an integrated biotechnology company, focuses on development and commercialization of late-stage therapeutics to improve health outcomes in treatment of cancer and other serious diseases. The company believes in delivering products and supporting medical teams with the purpose of achieving superior outcomes for patients and their families. Visit www.partnertx.com.
SOURCE Partner Therapeutics, Inc.
These press releases may also interest you
|
News published on and distributed by: