Subject: FDA
Innovative Aspiration Thrombectomy System by Expanse ICE Receives FDA Clearance for vessels of the peripheral arterial and venous systems
PLEASANTON, Calif., April 22, 2024 /PRNewswire/ -- Expanse ICE announced today the ICE Aspiration System has received 510(k) clearance from the U.S. Food and Drug Administration. This announcement introduces a new and exciting player in the peripheral thrombectomy market.
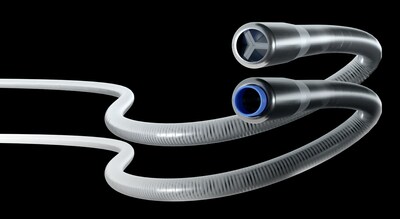
The ICE System is specifically designed to address the complex challenges associated with peripheral thrombectomies. Blood clots are the third most common vascular disease. Almost one million patients suffer from peripheral blood clots that must be treated each year, and up to 33% of them suffer long-term complications, according to the Center for Disease Control.
Eitan Konstantino, PhD, a serial entrepreneur in the vascular device field, is the driving force behind Expanse ICE. "We've engineered the ICE catheter system to harness the aspiration power typical of a large bore catheter, but within the slender profile of a much smaller device," Dr. Konstantino said.
"It is clear this device was built with physicians in mind. It aims to address some of the biggest issues we see regularly. The device was designed and built with a strong understanding of foundational physics that leaves me optimistic for its success," said Dr. Michael Lichtenberg, Chief Medical Officer of Angiology at the Vascular Center Clinic in Arnsberg, Germany.
"I am looking forward to being among the first users of the Expanse ICE system. Its ability to offer powerful clot removal in a compact size is an intriguing proposition that could significantly enhance our ability to meet patient needs more effectively and improve treatment outcomes in peripheral vascular disease," said Dr. Aravinda Nanjundappa, Director of Peripheral Vascular Interventions, at the Cleveland Clinic.
"The thrombectomy market is witnessing robust growth and the approval of the ICE system comes at an auspicious time. It is cutting-edge technology in an underpenetrated market," said Shlomi Nachman, former Johnson & Johnson Company Group Chairman of Cardiovascular & Specialty Solutions and Vision.
About Expanse ICE
Expanse ICE is a company born from Expanse Medical, a medical device incubator founded by Dr. Konstantino. The team responsible for the Expanse ICE system also built AngioSculpt, Chocolate and a variety of neurovascular products. Expanse Medical is focused on developing solutions for vascular diseases. The company is dedicated to enhancing the quality of patient care through technological advancements and rigorous research.
SOURCE Expanse Medical
These press releases may also interest you
|
News published on and distributed by: