Subjects: MAV, RCL
Advisory - Two lots of Advil Cold & Sinus Day/Night Convenience Pack recalled due to labelling error that may cause confusion between daytime and nighttime tablets
OTTAWA, ON, Oct. 3, 2021 /CNW/ -
Summary
- Product: Advil Cold & Sinus Day/Night Convenience Pack, in boxes of 18 caplets and 36 caplets
- Issue: GSK is recalling two lots (ER2072, ER2069, expiry 2023-02) due to a labelling error on the back of the blister pack. Consumers may take a nighttime caplet instead of a daytime caplet, and vice versa. Taking a nighttime caplet when alertness is required may pose health risks.
- What to do: Stop using the recalled products. Consult a health care professional if you have used any of these products and have health concerns.
Issue
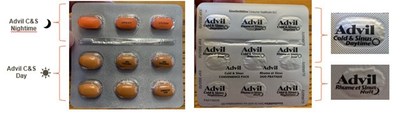
GlaxoSmithKline Consumer Healthcare ULC (GSK) is recalling two lots of Advil Cold & Sinus Day/Night Convenience Pack (one lot of 18 caplet boxes and one lot of 36 caplet boxes) due to a labelling error on blister packs. The foil backing on the blister pack is rotated upside down and misaligned, so the nighttime caplets are labelled as daytime caplets, and some daytime caplets are labelled as nighttime caplets. Consumers may take a nighttime caplet when they intend to take a daytime caplet, and vice versa.
The daytime and nighttime caplets have a different shape and colour (the daytime caplet is beige and has the marking 'ADVIL COLD & SINUS' in black ink. The nighttime caplet is orange and has the marking 'Advil A/S'). The nighttime caplets contain an antihistamine medicinal ingredient (chlorpheniramine maleate) that can cause drowsiness. Taking a nighttime caplet when alertness is required may pose potentially serious adverse health consequences, such as when driving motor vehicles or operating heavy machinery. It may also cause potentially serious health consequences for those who have taken other sedatives or tranquilizers, consumed alcohol, and the elderly.
The affected products were distributed in Canada starting July 2021.
Health Canada is monitoring the company's recall and implementation of any necessary corrective and preventive actions.
Affected products
Product Name | DIN | Lot # | Expiration Date |
Advil Cold & Sinus Day/Night Convenience Pack Box of 36 caplets (24 daytime and 12 nighttime) | 02399733 (convenience pack) 02267616 (daytime caplets) 02267632 (nighttime caplets) | ER2072 | 2023-02 |
Advil Cold & Sinus Day/Night Convenience Pack Box of 18 caplets (12 daytime and 6 nighttime) | ER2069 | 2023-02 |
What you should do
- Stop using the recalled products. Consult a health care professional if you have used any of these products and have health concerns.
- Follow municipal or regional guidelines on how to dispose of chemicals and other hazardous waste; or
- Return the product to your local pharmacy for proper disposal.
- Contact GSK by calling 1-855-367-7349, or emailing [email protected], if you have questions about the recall.
- Report any health product-related side effects or complaints to Health Canada.
Images
Incorrectly labelled Advil Cold & Sinus Day/Night Convenience Pack blister pack, front and back. The top row contains orange nighttime caplets, followed by two rows of beige daytime caplets. The foil backing on the blister pack is rotated upside down and misaligned, so the nighttime caplets are labelled as daytime caplets, and some daytime caplets are labelled as nighttime caplets.
SOURCE Health Canada
These press releases may also interest you
|
News published on and distributed by: