Subjects: RCL, CFG
Advisory - Multiple cold and flu powdered medications, for adults and children 12 years of age and older, recalled due to potential health risks
OTTAWA, ON, Dec. 20, 2021 /CNW/ -
Summary
- Product: Multiple generic store-brand cold and flu powdered medications for adults and children 12 years of age and older.
- Issue: CellChem Pharmaceuticals Inc. is recalling all lots of several products due to potential health risks. The company could not demonstrate that products remain safe and of good quality until their expiry date, and multiple lots had inconsistent quantities of active ingredients in the products.
- What to do: Stop using the recalled products. Talk to your healthcare provider if you or your child have taken a recalled product and have health concerns.
Issue
CellChem Pharmaceuticals Inc. is recalling all lots of cold and flu medications, sold in pouches of dissolvable powder, due to potential health risks. The products are authorized for use in adults and children 12 years of age and older. They are available over-the-counter and sold under various generic store-brand labels at many retailers across Canada.
The products are being recalled because the company could not demonstrate that products remain safe and of good quality until the expiry date. In addition, multiple lots had active ingredients, such as acetaminophen, that were not in the amounts listed on the product label.
Products that contain less than the labelled quantity of active ingredients may be less effective. Taking products that contain more than the labelled quantity of active ingredients can lead to inadvertently exceeding the maximum daily doses. For example, products that contain too much acetaminophen could pose serious health risks, such as liver damage. Signs of taking too much acetaminophen include nausea, vomiting, lethargy, sweating, loss of appetite and pain in the upper part of the abdomen or stomach. Abdominal pain may be the first sign of liver damage and may not be apparent for 24 to 48 hours. Risks of taking too much of an active ingredient may be higher for children and adolescents.
Health Canada identified these issues during an inspection. As a result, the company initiated a recall in September 2021 of certain powdered cold and flu medications. This recall is now being expanded after further investigation by the company identified additional affected products.
At Health Canada's direction, CellChem Pharmaceuticals Inc. has stopped selling and is recalling the affected products. Health Canada is monitoring the company's recall and the implementation of any corrective and preventative actions. If additional safety concerns are identified, Health Canada will take appropriate action and inform Canadians as needed.
Affected products
Product | Generic Store Brand | DIN |
Extra Strength Total |
| 02420937 |
Hot Lemon Relief for
|
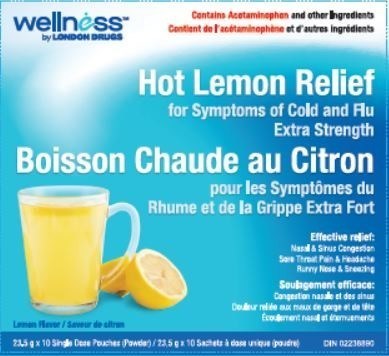
| 02238890 |
Hot Lemon Relief for
|
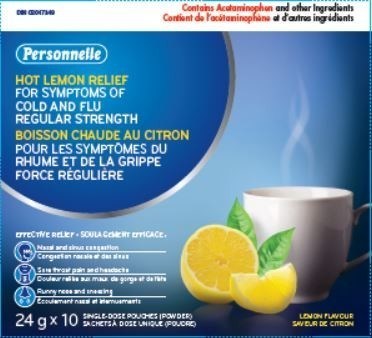
| 02047349 |
Hot Lemon Relief for |
| 02246848 |
Night Time Total Extra |
| 02344548 |
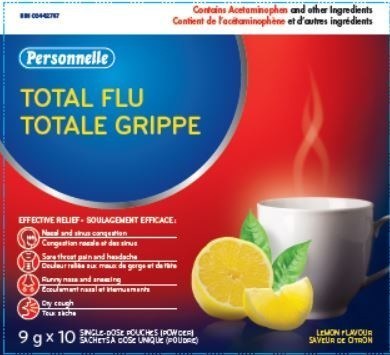
Total Flu
|
| 02442787 |
What you should do
- Stop using the recalled products. Talk to your healthcare provider if you or your child have taken a recalled product and have health concerns.
- Follow municipal or regional guidelines on how to dispose of chemicals and other hazardous waste; or
- Return the product to your local pharmacy for proper disposal.
- Contact CellChem Pharmaceuticals Inc. by calling 1-844-481-8884, or emailing [email protected], if you have questions about the recall.
- Report any health product-related side effects or complaints to Health Canada.
Également disponible en français
SOURCE Health Canada
These press releases may also interest you
|
News published on and distributed by: